Get Tech Tips
Subscribe to free tech tips.
Work Vehicle Entropy

Have you ever noticed that the more you're required to speed up to get all your work done in a day, the more the cleanliness of your work vehicle suffers?
Some techs won't clean their vans, no matter how slow or busy the schedule gets, but most of us prefer a clean and organized vehicle. However, our van cleanliness can slip a bit in the really busy season.
This is entropy at work.
One way to think of entropy is the tendency for energy to be “wasted” or for energy to become more disorganized as it is changed from one form to another. In practical terms, the higher the entropy, the greater the waste in moving energy around. The lower the entropy, the less energy is wasted or unusable.
When your van is clean, you waste less energy trying to find what you need, but that is easier to do when you aren't “rushed.”
Don't confuse entropy with enthalpy. Entropy is the measure of the “availability” of energy to do useful work; enthalpy is a measure of total heat content.
Alternatively, you can think of entropy as the tendency for everything to go from more organized to less organized over time.
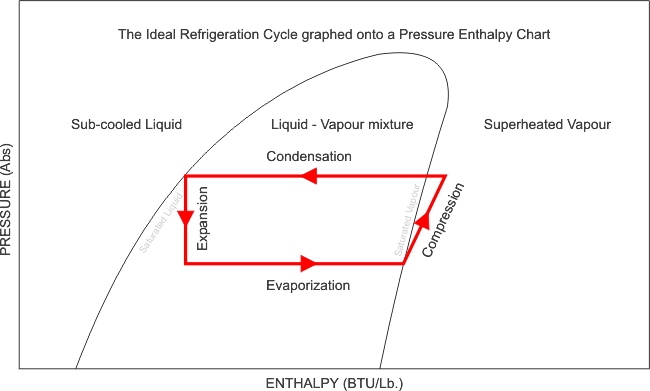
When we talk about entropy in classroom settings, we typically do it when analyzing a pressure-enthalpy diagram of a refrigeration circuit like the one shown above. You will notice the red shape is nearly a rectangle, barring the slanted line on the right; that line shows what happens when the refrigerant is compressed. Rather than going straight up and down, it curves to the right to show that additional heat is added to the refrigerant (enthalpy) as it is compressed. This increase in enthalpy follows something called lines of constant entropy; in other words, as more energy is added to a system, the faster the molecules move and the less organized they become.
As pressure and temperature increase, so (generally) does entropy. When it gets hot and the dispatcher starts putting pressure on you, your van entropy also increases.
In a refrigeration circuit, the system will work more efficiently when we achieve the desired movement of BTUs with a minimal amount of entropy. Practically speaking, this means using lower pressures and temperatures to move heat from one place to another when possible, but we still need to get the job of moving heat from one place to another done. In that process, entropy WILL occur.
We all know the guy who has the PERFECT van, the spotless tools, and the clean uniform. His van stays that way because he hardly does anything, and then he takes 20 minutes after every call, wiping down and oiling his tools. He has very little entropy in his van because there is very little WORK being done.
Absolute zero is the temperature at which no molecular motion exists. In that state, there is also no entropy. No heat, no work, no disorganization. More heat, more work, more disorganization (entropy).
So, don't use me as an excuse for a messy van, but if your boss hassles you too much on a busy day, you can remind him of what we discussed today. Heat and pressure result in entropy, and the back of that van—that's entropy, baby.
—Bryan
P.S. – I know that this article took some liberty with definitions. It's an analogy, not a science textbook. You can find another explanation of entropy HERE.










Comments
Brilliant article and concept! It takes less energy to keep your work vehicle clean and tidy than to try and find what you’re looking for on the job if left disorganized. That’s entropy baby!
Brilliant article and concept! It takes less energy to keep your work vehicle clean and tidy than to try and find what you’re looking for on the job if left disorganized. That’s entropy baby!
pharmacie en ligne livraison europe: pharmacies en ligne certifiГ©es – Pharmacie sans ordonnance pharmafst.com
pharmacie en ligne livraison europe: pharmacies en ligne certifiГ©es – Pharmacie sans ordonnance pharmafst.com
http://pharmafst.com/# pharmacie en ligne france fiable
http://pharmafst.com/# pharmacie en ligne france fiable
pharmacie en ligne france pas cher pharmacie en ligne avec ordonnance or pharmacie en ligne pas cher
https://images.google.ee/url?sa=t&url=https://pharmafst.com pharmacie en ligne pas cher
[url=https://www.google.com.bz/url?sa=t&url=https://pharmafst.com]Achat mГ©dicament en ligne fiable[/url] pharmacie en ligne pas cher and [url=http://www.1moli.top/home.php?mod=space&uid=111836]Pharmacie sans ordonnance[/url] pharmacie en ligne pas cher
pharmacie en ligne france pas cher pharmacie en ligne avec ordonnance or pharmacie en ligne pas cher
https://images.google.ee/url?sa=t&url=https://pharmafst.com pharmacie en ligne pas cher
[url=https://www.google.com.bz/url?sa=t&url=https://pharmafst.com]Achat mГ©dicament en ligne fiable[/url] pharmacie en ligne pas cher and [url=http://www.1moli.top/home.php?mod=space&uid=111836]Pharmacie sans ordonnance[/url] pharmacie en ligne pas cher
pharmacie en ligne sans ordonnance [url=https://pharmafst.shop/#]pharmacie en ligne[/url] pharmacie en ligne livraison europe pharmafst.shop
pharmacie en ligne sans ordonnance [url=https://pharmafst.shop/#]pharmacie en ligne[/url] pharmacie en ligne livraison europe pharmafst.shop
Kamagra pharmacie en ligne: Acheter Kamagra site fiable – kamagra gel
Kamagra pharmacie en ligne: Acheter Kamagra site fiable – kamagra gel
Cialis sans ordonnance pas cher Tadalafil sans ordonnance en ligne or Cialis generique prix
http://new.0points.com/wp/wp-content/plugins/wp-js-external-link-info/redirect.php?url=https://tadalmed.com Tadalafil achat en ligne
[url=https://yassu.jp/bidan/town/kboard.cgi?mode=res_html&owner=proscar&url=tadalmed.com]Cialis sans ordonnance 24h[/url] Acheter Cialis 20 mg pas cher and [url=http://www.xgmoli.com/bbs/home.php?mod=space&uid=7038]cialis sans ordonnance[/url] Acheter Cialis 20 mg pas cher
Cialis sans ordonnance pas cher Tadalafil sans ordonnance en ligne or Cialis generique prix
http://new.0points.com/wp/wp-content/plugins/wp-js-external-link-info/redirect.php?url=https://tadalmed.com Tadalafil achat en ligne
[url=https://yassu.jp/bidan/town/kboard.cgi?mode=res_html&owner=proscar&url=tadalmed.com]Cialis sans ordonnance 24h[/url] Acheter Cialis 20 mg pas cher and [url=http://www.xgmoli.com/bbs/home.php?mod=space&uid=7038]cialis sans ordonnance[/url] Acheter Cialis 20 mg pas cher
kamagra livraison 24h [url=https://kamagraprix.com/#]kamagra pas cher[/url] kamagra 100mg prix
kamagra livraison 24h [url=https://kamagraprix.com/#]kamagra pas cher[/url] kamagra 100mg prix
olympe: olympe casino cresus – olympe casino avis
olympe: olympe casino cresus – olympe casino avis
I’m really inspired with your writing abilities and also with the structure on your blog. Is this a paid subject matter or did you customize it yourself? Either way stay up the excellent quality writing, it is rare to see a nice blog like this one these days!
I’m really inspired with your writing abilities and also with the structure on your blog. Is this a paid subject matter or did you customize it yourself? Either way stay up the excellent quality writing, it is rare to see a nice blog like this one these days!
To leave a comment, you need to log in.
Log In